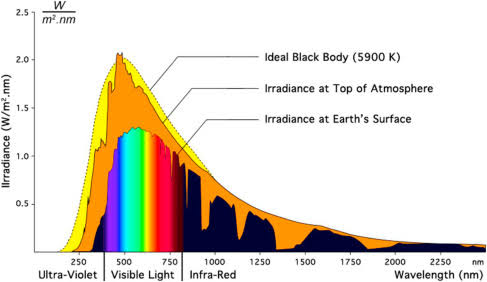
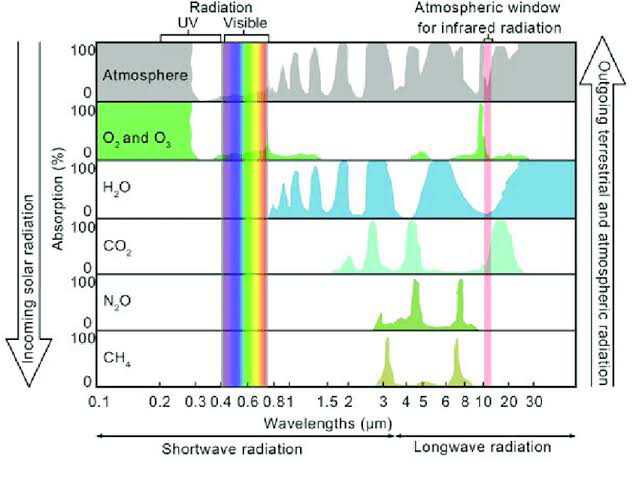
I came up with a pretty straightforward way of debunking the “greenhouse effect” in relation to carbon dioxide and the atmosphere.
It’s a 1 dimensional example, but it could be expanded into 2 and 3 dimensions.
It works well in a spreadsheet, but can be described pretty well.
If you can imagine a line with the sun on the left, the atmosphere in the middle, and earth on the right, one can put arbitrary numbers to that.
We calculate only a single “instant”.
We assign the middle number an absorption value, and the earth the same.
So the start of the “instant” looks like this;
From left to right, the sun (100), the energy emitted from the “greenhouse gas” going back into space(0), the “greenhouse gas” (0), the energy emitted towards the earth (0), and the earth (0).
We can arbitrarily assign the “greenhouse gas” an absorption of 50% and emission value of 100%. That is, half the energy is absorbed, half passes through.
Note the sun will always be 100. It doesn’t get anything back.
All lines (passes) are the same “instant”, and the energy is not renewed from the sun.
Beginning state;
100 0. 0. 0. 0.
First pass, the 100 has been half absorbed by the gas, half passes through to the earth.
100 0. 50. 0. 50.
Pass two, the gas emits 100% of it energy in both directions.
100 25 0 25 50
Third pass, the earth has now all the initial energy. We arbitrarily say it’s going to emit 100% back from right to left in Pass 4.
100 0 0 0 75
Pass 4, the energy is now in the gas.
100 0 75 0 0
Pass 5
- 37.5. 0 37.5 0
Pass 6
-
-
-
- 37.5
And so on. The energy from the gas to the earth will trend towards zero.
We can keep running passes on this instant.
If we now increase the density of the gas, so it now absorbs 60% of the energy, something quite obvious happens
100 0. 0. 0. 0
-
- 60 0. 40
100 30 0 30. 40
-
-
-
-
- Note this number! 70!
At the same point in the previous example, we had 75!
So, if we increase the gas absorption and emits 100% of that energy, the "earth "
got cooler, not hotter.
It doesn’t matter if we now take our figures and run a “second instant”, for example,
-
-
-
- 37.5
Running the same process will always result in the right hand number being smaller if we increase the absorption value of the middle number.
In short, as this is really just a 1D example, there is no way around the obvious, any increase in absorption of a gas (or any medium) will result in less heat on the other side of it.
![]() .
.